The bio-accumulation of fluoride: a response to NHMRC
In 2018 the CEO of Australia’s National Health and Medical Research Council publicly advised that fluoride is not bio-accumulative, stating that it “achieves a ‘steady state’ that is safe for humans”. Many scientific studies over the last 50 years show that our fluoride intake is only partially excreted and that it accumulates in tissues, particularly bone. It is astounding that the head of our national health body or her “expert” advisers can be so ignorant on this important health matter.


- How can the head of our national health body and her “experts” make such erroneous statements and mislead key decision makers such as Australian councils?
- An FOI request to NHMRC returned four scientific references that only confirm fluoridation chemicals are in fact bioaccumulative;
- Numerous additional studies also show that fluoride accumulates in the human body;
- Fluoride accumulates at much higher rates in young children – around 80% accumulates in their young, developing bones;
- Like the UK, Australia and New Zealand are are large tea-drinking countries. Black tea is often high in fluoride;
- There are other vulnerable sub-population groups such as the elderly, diabetics, other people with impaired kidney function, those hypersensitive to fluoridation chemicals or those consuming extra-high amounts of water such as sportspeople and outdoor labourers. How can these subgroups continue to be ignored?
- The Weatherall study (1975) is very relevant to Australia and to this subject and yet was arrogantly dismissed by NHMRC;
- The half-lives of lead and fluoride are in fact similar;
- Given the daily exposure to fluoride, most people are likely to accumulate fluoride, unlike lead.
Ignorance or deception?
It would be more reassuring if it was only Professor Anne Kelso, CEO of Australia’s National Health and Medical Research Council (NHMRC), who does not understand the science and technical aspects of fluoride metabolism and toxicokinetics. At the very least it is obvious that the verbal explanations given to Professor Kelso by her “experts” were either misunderstood or suggest the experts are as ignorant of the meaning of bioaccumulation as their CEO. If so, this level of professional incompetence is very disappointing from toxicologists employed by the Australian government.
But in this case, ‘ignorance’ and ‘professional incompetence’ might be lenient descriptions. Given the considerable public resources at Anne Kelso’s disposal as CEO of NHMRC, including receipt of substantial correspondence and detailed submissions opposed to the use of fluoridation chemcials (which have encompassed the subject of bioaccumulation), it is difficult to understand how Ms Kelso and her expert advisers could be ignorant of this matter.
An open letter to all councillors
On 8 March 2018, following a presentation the previous day to Councillors of the NSW town of Oberon, NHMRC’s CEO wrote an open letter (“Open Letter”) addressed to all Councillors in Australia. The presentation, given by four professionals, highlighted the lack of safety, lack of efficacy and illegal nature of water fluoridation.
Prof. Kelso’s Open Letter assures councillors that “NHMRC found no reliable evidence that community water fluoridation at current Australian levels causes health problems”. The letter discloses that this assurance is based on NHMRC’s own review dated November 2017, but omits to mention that the review in question considers only selected texts published between 2006 and 2015, with many irrational exclusions and exceptions according to NHMRC’s heavily-biased fluoride-promoting agenda. (More details of the questionable methodology utilised by NHMRC are provided in the article; Can the NHMRC be trusted?)
In addition, Professor Kelso also stated:
“Some people ask if there is a cumulative effect of consuming fluoride over a lifetime. The toxicologists on our expert committee advised that fluoride is excreted regularly by the kidneys to achieve a ‘steady state’ that is safe for humans. This is different to lead which accumulates in the body.”
An excerpt from an open letter to Councillors from Professor Anne Kelso, CEO, NHMRC, 8 March 2018.
These incorrect statements, along with NHRMC’s response to a request for additional information specifically about this subject and subsequent comments of clarification made by Professor Kelso and her staff, should be of great concern to all state governments who are ultimately responsible for the process of fluoridating public water supplies.
A request for more information
When asked to provide any published or unpublished scientific research studies (via a Freedom of Information request) to back up the statement that fluoride is not bioaccumulative, NHMRC stated “The advice on toxicity and whether or not fluoride bio accumulates was provided verbally in a discussion during a Fluoride Reference Group meeting…” and “the toxicologists provided advice in the course of advising on edits to the draft Information Paper”.
Before going any further, we should point out that, despite these apparent discussions, there is no mention of the bioaccumulation or elimination of fluoride in the 95-page Information Paper (2017), referred to above.
In their FOI response, NHMRC provided some “citations of interest”, which are apparently supposed to support their claim; however seem to do the opposite. These four citations are as follows:
- European Food Safety (EFSA) Panel on Dietetic Products, Nutrition and Allergies, 2013. Scientific opinion on Dietary Reference Values for fluoride.
Within this document, Section 2.3.5 on Elimination (page 13), states:
“Absorbed fluoride which is not deposited in calcified tissue is mainly excreted via the kidney (around 60% in adults, 45% in children) (Villa et al., 2010). The percentage of absorbed fluoride excreted via the kidney in infants and young children can be as low as 10-20 % because of a higher capacity of bone to accumulate fluoride.”
In other words, NHMRC has referred to a study confirming that young children can accumulate around 80 to 90 per cent of the fluoride they ingest from various sources, including water fluoridation; that normal, healthy children may accumulate around 55 per cent; and normal, healthy adults may accumulate around 40 per cent.
These accumulation rates, however, can increase significantly for disadvantaged subpopulation groups. See the sections below on “Infants and children” and “Other subpopulation vulnerability”; however even the Australian Drinking Water Guidelines (2011), states: “People with kidney impairment have a lower margin of safety of fluoride intake. Limited data suggests that their fluoride retention may be up to three times normal.”
2. National Research Council (2006). Fluoride in Drinking Water: A Scientific Review of EPA’s Standards. Washington, DC: The National Academies Press.
This thorough 318-page review, conducted over three years by a panel of 12 scientists and health professionals, made several references to the accumulation of fluoride, as follows:
“Fluoride is readily incorporated into the crystalline structure of bone and will accumulate over time.” (p 4)
“The models estimated that bone fluoride concentrations resulting from lifetime exposure to fluoride in drinking water at 2 mg/L (4,000 to 5,000 mg/kg ash) or 4 mg/L (10,000 to 12,000 mg/kg ash) fall within or exceed the ranges historically associated with stage II and stage III skeletal fluorosis (4,300 to 9,200 mg/kg ash and 4,200 to 12,700 mg/kg ash, respectively).” (p 5)
“Chronic dosing leads to accumulation in bone and plasma” (p 76)
“Both models predicted increasing fluoride concentrations in bone with length of chronic exposure.” (p 76)
“Subsequent decreases in exposure cause fluoride to move back out of bone into body fluids, becoming subject to the same kinetics as newly absorbed fluoride.” (p 76)
“Several cross-sectional studies have found an association between fluoride bone concentrations and age (Jackson and Weidmann 1958; Kuo and Stamm 1974; Parkins et al. 1974; Charen et al. 1979; Alhava et al. 1980; Eble et al. 1992; Richards et al. 1994; Torra et al. 1998).” (p 77)
“Bone fluoride concentrations increase with both magnitude and length of exposure.” (p 82)
“The prolonged maintenance of fluoride in the bone requires that uptake of the element occurs at the same or greater rate than its clearance. This appears to be the case. (See Chapter 3 for more detailed discussion of the pharmacokinetic data on fluoride.)” (p 108)
“Hence, it is reasonable that 99% of the fluoride in humans resides in bone and the whole body half-life, once in bone, is approximately 20 years (see Chapter 3 for more discussion of pharmacokinetic models).” (p 108)
3. SCHER, Opinion on critical review of any new evidence on the hazard profile, health effects, and human exposure to fluoride and the fluoridating agents of drinking water; May 2011.
There is little mention in this reference regarding the accumulation or elimination of fluoride; however the following statements may be relevant:
“Concerns regarding the potential carcinogenic effect of fluoride have been focused on bone cancer due to the known accumulation of fluoride in bones. Osteosarcoma is a rare form of cancer making it difficult to analyse risk factors using epidemiology.” (p 16)
“There are no apparent age related differences in renal clearance rates (adjusted for body weight or surface area) between children and adults. However, in older adults (more than 65 years of age), a significant decline in renal clearance of fluoride has been reported consistent with the age-related decline in glomerular filtration rates.” (p 12)
“Approximately 99% of the fluoride in the human body is found in bones and teeth. Fluoride is incorporated into tooth and bone by replacing the hydroxyl ion in hydroxyapatite to form fluorohydroxyapatite. The level of fluoride in bone is influenced by several factors including age, past and present fluoride intake, and the rate of bone turnover. Fluoride is not irreversibly bound to bone and is mobilized from bone through bone remodelling.” (p 12)
4. Whitford GM (1996). The metabolism and toxicity of fluoride. Monogr Oral Sci. 1996.
“The quantitatively important fates of absorbed fluoride are uptake by calcified tissues and excretion in the urine. Roughly 50% of an absorbed amount will be excreted in the urine during the following 24h while most of the remainder will become associated with calcified tissue. These fractions, however, can vary widely depending on several variables as will be discussed later.” (p 1)
“The peak level usually occurs during the first hour after ingestion. After the bulk of the dose has been absorbed, the plasma levels show a rapid decline due to the continuing uptake by bone and urinary excretion.” (p 3)
Therefore, in response to very specific requests to provide any reference material to substantiate the claim that fluoride is not bioaccumulative, NHMRC has in fact only provided references that confirm the opposite – that fluoride does indeed accumulate in the body. They also ignore that rates of bioaccumulation will vary significantly according to the health and demographic circumstances of each individual. See section below on infants and other subpopulation vulnerability.
Additional science on bioaccumulation
In 1971, Marier & Rose1 stated “Healthy adult kidneys excrete 50 to 60% of the fluoride ingested each day”
In 1975, Weatherell et al published a study titled “Fluoride and its Relation to Bone and Tooth”2. Also referring to other previous studies, the authors concluded:
“Unless the ingested fluoride is in an insoluble form (ERICSSON, 1958; WEDDLE and MUHLER, 1957) it is quickly absorbed (HODGE, 1956) and although excretion via the kidney is very efficient, up to half the absorbed fluoride is incorporated into the skeleton, where it accumulates with time (LARGENT and HEYROTH, 1949). In many experimental studies using relatively large dosage of fluoride, it was found that the initial rapid rate of incorporation into the skeleton gradually decreased, presumably due to saturation of the sites of skeletal uptake (ZIPKIN and McCLURE, 1952; WEATHERELL, 1969). At the restricted concentrations present in human diets, this does not occur and throughout life the concentration of fluoride in bone progressively increases (WEATHERELL, 1966). The bone of older individuals therefore contains a relatively high concentration of fluoride. Even in Leeds, where the concentration in the water supply is low (<0.1 ppm), the cortical bone in femora from elderly individuals can contain almost 3,000 ppm F on an ash basis. (Fig. 1.)”

Therefore, according to Weatherall et al, fluoride ingested by residents of Leeds, England, steadily accumulates in the skeletal system over a lifetime and as a result, the elderly may have an extremely high bone-fluoride concentration of around 3,000ppm. It should be noted that the drinking water in Leeds contains less than 0.1mg of fluoride per litre (around 10% of the fluoride concentration in Australia); however the residents ingest fluoride from tea and other dietary sources. This source of fluoride makes this study particularly relevant and applicable, because Australia – like England – is a nation of tea drinkers.
In 2016, a study conducted by Roy Morgan Research3, revealed that average tea consumption in Australia in a 12-month period in 2015 – 2016 was 9.5 cups per week. This ranged from 5.5 cups per week amongst teenagers aged 14 to 17 years, up to 10.9 cups for those aged 65 and above. Only tap water, milk and coffee are more widely consumed and coffee is often consumed using fluoridated tap water.
As for the fluoride content in tea, one study conducted in New Zealand (which has similar tea brands as Australia) by environmental scientist Declan Waugh4, found that fluoride content in the 18 most common brands ranges from 0.4 to 3.2mg/L for each tea bag, when using unfluoridated water and 0.9 to 3.9mg/L when using fluoridated water containing 0.7mg/L of fluoride.

Then there’s the fluoride from other sources, such as toothpaste, processed foods, various medications and pesticide residue on fruit and vegetables – taking the average fluoride dosage for Australians significantly higher than the population of Leeds.
In 1997 and 2001, Luke5 noted that fluoride accumulates in the body, largely in calcifying tissues such as the bones and pineal gland.
“After half a century of the prophylactic use of fluorides in dentistry, we now know that fluoride readily accumulates in the human pineal gland. In fact, the aged pineal contains more fluoride than any other normal soft tissue.”
Infants and children
Several studies have demonstrated that infants and young children in particular are less able to process and excrete toxic fluoridation chemicals and therefore their rates of accumulation are significantly higher than adults.
“Approximately 80% of an absorbed dose of fluoride is retained in young children compared to 50% in adults. This is supported by the finding that renal fluoride excretion rate is lower in children than adults. This difference in fluoride retention is due to high fluoride uptake in developing bones.” Whitford GM. (1994).6
“Overall, an average of 86.8% of the dose was retained by the infants, which is about 50% higher than would be expected for adults… There is a clear need for more information about the renal handling and general metabolism of fluoride in young children…” Ekstrand J, et al. (1994).7
“Our finding that mean retention of fluoride [among infants] was 83.4% of the quantity of fluoride absorbed from the dose of supplement may be compared with the finding in adults that mean extrarenal clearance of fluoride from a dose of supplement given to adults was 55.3%.” Ekstrand J, et al. (1994).8
“The breast-fed infants were in a negative F balance, excreting more F than they ingested. The bottle-fed infants, on the other hand, retained more than 50% of the ingested F dose. The rapidly growing skeleton certainly enhances F retention, but a contributing factor might also be a slow elimination rate for F, caused by a not fully developed renal handling capacity for F at this early stage of life.” Ekstrand J, et al. (1984).9
“… in weaned infants, a relatively large proportion of fluoride intake is retained in the body (i.e. deposited in calcifying tissues).” (Zohoori et al 2019)10
Other subpopulation vulnerability
There are other subgroups in our community who will experience increased accumulation of fluoride. Individuals in these subgroups are therefore more vulnerable to the short-term and long-term toxic effects of fluoridation chemicals.
Apart from infants and children, these include the elderly, diabetics, other people with impaired kidney function, those with pre-diabetic or pre-renal-function impairment, those with poor nutritional status or who suffer from malnutrition (e.g. deficient in magnesium, vitamin C, vitamin D and iodine), those with heightened sensitivity to fluoride and many people with high or extra-high water consumption such as sportspeople and outdoor labourers.
The bioaccumulation of lead vs fluoride
Professor Kelso states that fluoride “achieves a ‘steady state’ that is safe for humans”, and “This is different to lead which accumulates in the body.” This last sentence is also incorrect and deserves specific attention.
Like fluoride, most lead retained in the human body is ultimately deposited in the bones. According to the US Agency for Toxic Substances and Disease Registry (ATSDR); “The bones and teeth of adults contain about 94% of their total lead body burden; in children, that figure is approximately 73% [Barry 1975, as cited in ATSDR 2010].”11
Although the plasma (blood) elimination half-life of fluoride is much shorter than lead, the bone elimination rates for both tocix substances are more relevant. The elimination half-life of lead in cortical bone is estimated at 10 to 30 years12. For fluoride, according to the NRC 2006 review, the half-life elimination of fluoride from bone is estimated at 20 years. So their skeletal bioaccumulation rates are actually similar.
However, there is a vitally important distinction to be made: the typical exposure histories of both toxins are crucial. Fluoridation chemicals added to drinking water, along with other sources of fluoride such as processed foods, tea and toothpaste, will cause a constant continuous exposure over an entire lifetime, thus causing a steadily increasing bioaccumulation in the body (mostly the skeleton). In contrast, lead exposures for the majority of people are more episodic, occurring mostly in childhood. So for many people, their skeletal lead bioaccumulation will taper off after childhood. It may also be more common for a net removal of lead from the skeleton, at least in adults. In contrast, for anyone consuming fluoridated water and fluoride from other sources, it would be unlikely for them to have a net outflow of fluoride from their body. This directly contradicts what professor Kelso has stated.
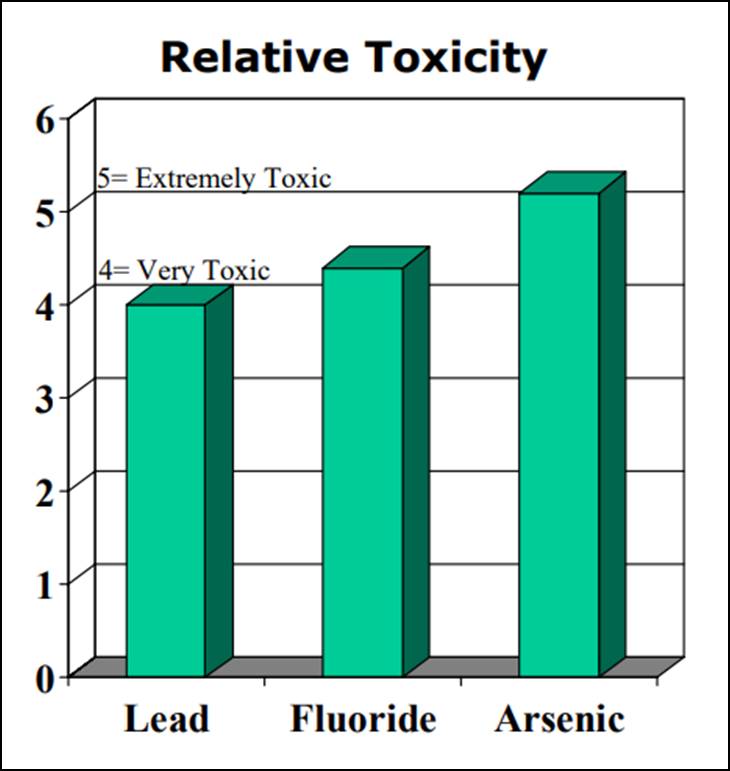
It should also be noted that, according to the “Clinical Toxicology of Commercial Products”13, fluoride is more toxic than lead and slightly less toxic than arsenic. (see chart below)
For more details on the history of lead and the similarities to fluoride, see our article “Fluoride is a neurotoxin”.

Subsequent events
In a letter dated 25 October 2018, Mr Alan Singh, Executive Director or NHMRC’s Translation Brach, responded to further concerns regarding the bioaccumulation of fluoride. Specifically addressing the Weatherell study1, Singh stated that he regarded “the Weatherell chapter as reputable, but not specific to the Australian context, which was the focus of our enquiry”. Given the low concentration of fluoride in Leeds compared to Australia and the similarities in tea drinking habits (as mentioned above) it is difficult to see how this study is not extremely relevant and how this is not a feeble excuse.
On 9 November 2018, Professor Anne Kelso also responded to another letter, now stating as follows:
“The FRG is aware of the deposition of fluoride in bones however this is not a health effect. In addition, the FRG understands the toxicokinetic effects of fluoride at levels used for water fluoridation and at higher levels, for example, that exposure to high levels of fluoride (>4mg/L) over 10-20 years can result in skeletal fluorosis. The FRG agreed to add text to the DIP about bone deposition and that fluoride is readily excreted by the kidneys and achieves a steady state in the body (contrasting with lead which accumulates)”.
Firstly, these subsequent comments by Professor Kelso, effectively admitting she was wrong, were contained in a private letter. Ms Kelso has not rectified her previous incorrect comments made publicly or to all councillors to whom the original letter was adressed.
Secondly, once again, these comments show a lack of understanding from NHMRC’s CEO, or poor advice from her expert toxicologists. She is confusing bioaccumulation (which she now describes as “deposition in bones”) with health effects. The concentration of fluoride in the skeleton increases over time, with roughly 50 per cent of ingested fluoride being deposited in the skeleton and thereby bioaccumulating.
Ms Kelso does not seem to understand that when a toxic chemical bioaccumulates in a tissue, it does not only have the potential to cause harm in that tissue. For fluoride, it is not just skeletal fluorosis or weakened bones that are the toxic endpoints from accumulated fluoride. The higher bone fluoride concentrations that occur with increased bioaccumulation mean that the plasma fluoride level will also be elevated because there is exchange between the bone and plasma compartments and the kidneys cannot immediately excrete all of the fluoride that enters the plasma, either from ingestion or re-mobilization from the bone. The plasma fluoride level is what most target tissues will experience, and the average plasma fluoride level will increase with increased bone fluoride concentration.
An infant with low fluoride intake and low prenatal exposure will have very low bone fluoride concentrations. If that child is given infant formula mixed with fluoridated water and later drinks fluoridated water, the bone fluoride concentration will steadily increase over a lifetime. Even if that person stops drinking fluoridated water (and other sources of fluoride), it will take about 20 years for the fluoride that has accumulated in the skeleton to be reduced by half. Twenty years is the estimated half-life of fluoride in bone according to the NRC 2006 report. If an individual drinks fluoridated water there will be no steady state of fluoride in their body, there will be an increasing deposition in their skeleton, the tissue that contains 99 per cent of the fluoride in a body. By definition, this is bioaccumulation.
Finally, it is also worth noting (as we have noted in other articles), that NHMRC is “Australia’s peak funding body for medical research, with a budget of roughly $900 million a year”. And yet, despite this massive funding, despite enormous resources available to all staff and advisers and despite the substantial and growing body of scientific research showing that fluoridation is neither safe nor effective in reducing tooth decay (including a comprehensive list of material sent specifically to Professor Kelso), NHMRC has not conducted, funded or encouraged any harms-related clinical studies to investigate the safety of low-level, long-term ingestion of fluoridation chemcials, other than dental fluorosis. At best this would appear unprofessional, if not reckless, irresponsible and entirely irrational; unless of course NHMRC’s mission is simply to defend the ridiculous paradigm of public water fluoridation.
For more details on the specific harms of fluoride, please see the following:
Fluoridation Letters sent to the US FDA (95 Set Series)
Fluoride is not safe
Fluoride and our bones
Fluoride and the endocrine system
Fluoride and diabetes
Fluoride and dental fluorosis
Conclusion
In other articles, especially our feature article “Can the NHMRC be trusted”, we have highlighted the lack of professionalism and poor methodology utilised by our national health body as far as the ongoing review and assessment of water fluoridation is concerned. This article has emphasised the inaccurate and misleading statements made by Professor Kelso in her open letter to councillors in March 2018. These statements were serious enough, but for her advisers to show such ignorance on this subject and for Professor Kelso and her staff to make subsequent dismissive and inaccurate statements should be of great concern to all state governments who are ultimately responsible for the process of fluoridating our public water supplies.
As we have shown in this article, fluoridation chemicals are bioaccumulative at approximately 50 per cent for healthy individuals. This rate can increase significantly for a substantial subpopulation of our society, especially infants and children and anyone with impaired kidney function. All affected subpopulations must not be ignored or consistently treated as irrelevant. The half-lives of fluoride and lead are also similar, however given daily exposure to fluoride, the general population are likely to accumulate fluoride to a greater extent than lead.
As we have also demonstrated in other articles, fluoride not only accumulates in the body, it causes various harms. Amongst other damages, fluoride is neurotoxic and nephrotoxic. For a national health body to arrogantly dismiss all the science showing the harm and potential harm that low-level, long-term ingestion of fluoridation chemcials can cause and to consistently refuse to conduct or fund any harms-related studies is not only unfathomable it’s negligent.
Reference notes:
- Marier, J.R. and Rose, D. 1971. “Environmental Fluoride”. Nal. Res. Council, Canada.
- Weatherell J.A., Deutsch D., Robinson C. (1975) “Fluoride and its Relation to Bone and Tooth”. In: Kuhlencordt F., Kruse HP. (eds) “Calcium Metabolism, Bone and Metabolic Bone Diseases”. Springer, Berlin, Heidelberg (pp 101-102)
- Roy Morgan Research (2016). “The tea party: Australians love a cuppa”.
- Waugh , D.T. et al, (2017). “Black Tea Source, Production, and Consumption: Assessment of Health Risks of Fluoride Intake in New Zealand”. Journal of Environmental and Public Health.
- Jennifer Anne Luke, 1997 “The Effect of Fluoride on the Physiology of the Pineal Gland”. A dissertation submitted to the School of Biological Sciences, University of Surrey, in fulfilment of the requirements for the Degree of Doctor of Philosophy; and Jennifer Anne Luke, 2001 “Fluoride deposition in the aged human pineal gland”; School of Biological Sciences, University of Surrey, Guildford, UK.
- Agency for Toxic Substances & Disease Registry (ATSDR) (2003). “Toxicological profile for fluorides, hydrogen fluoride, and fluorine.” Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service.
- Whitford GM. (1994). “Intake and metabolism of fluoride”. Advances in Dental Research 8:5-14.
- Ekstrand J, et al. (1994). “Fluoride pharmacokinetics in infancy”. Pediatric Res. 35:157-63.
- Ekstrand J, et al. (1984). “Fluoride Balance Studies on Infants in a 1-ppm-Water-Fluoride Area.” Caries Res. 18:87-92.
- Fatemeh V. Zohoori et al, (November 2018). “Fluoride retention in infants living in fluoridated and non-fluoridated areas: effects of weaning.” British Journal of Nutrition.
- Agency for Toxicity and Disease Registry (2017). “Lead Toxicity: What is the Biological Fate of Lead in the Body?”
- Christopher P. Holstege MD, Medscape (2015). “Etiology of Lead Toxicity.”
- Gosselin, Robert E., Smith, Roger P., Hodge, Harold C., Brad (1984).; “Clinical Toxicology of Commercial Products” 5th Edition.